The MGuard(TM) Coronary's innovative concept has enjoyed an enthusiastic welcome from leading interventional cardiologists around the world. 
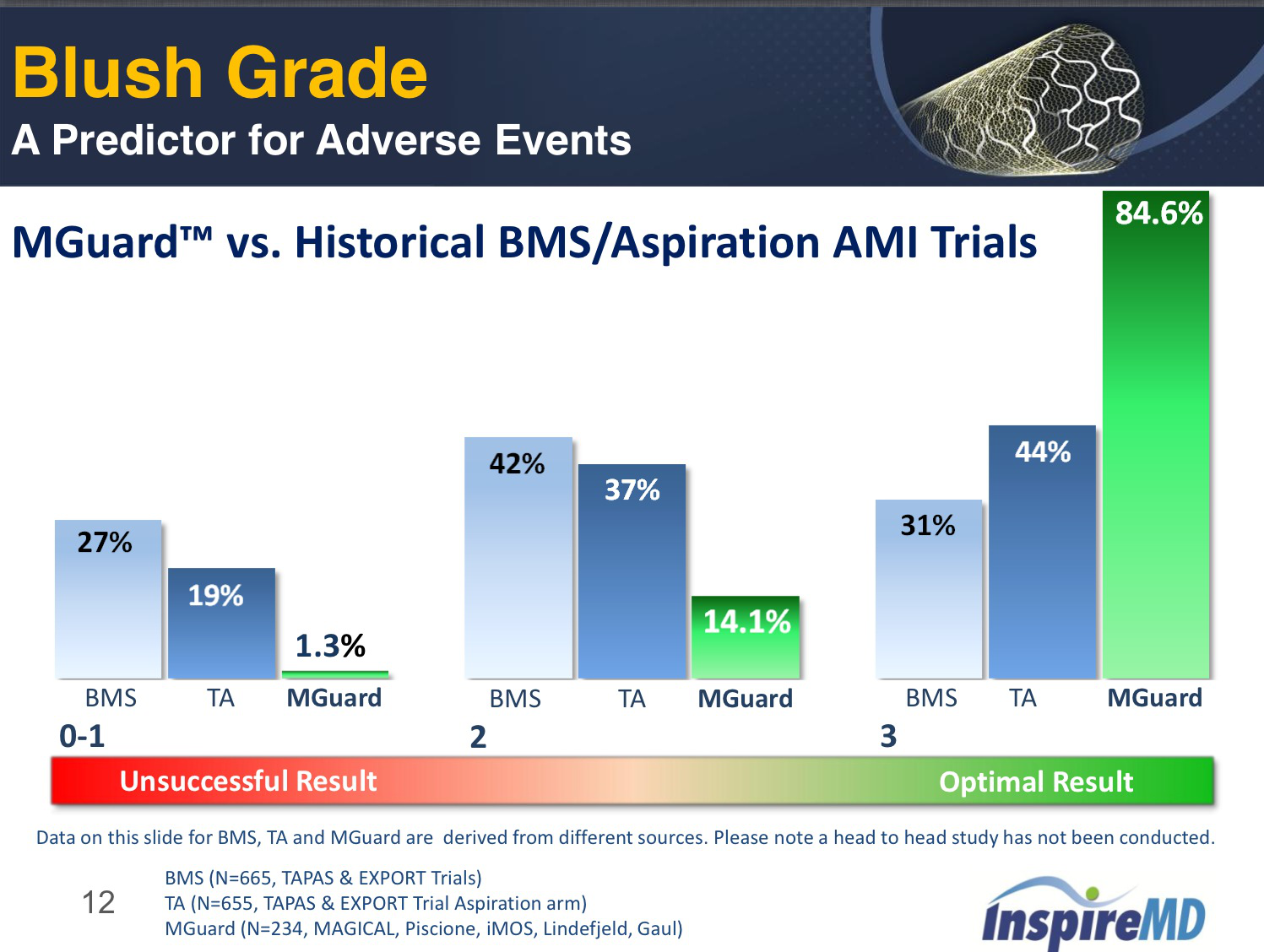
The sleeve is designed to diffuse stent pressure on the vessel wall, thereby may reduce injury and lower the likelihood of restenosis. The MGuard(TM) Coronary stent provides long acting embolic protection, without adding complexity in deliverability. The sleeve is designed to expand seamlessly when the stent is deployed, without affecting the structural integrity of the stent, and to prevent plaque detachment during and post procedure. The protective sleeve is composed of a micron-level-fiber knitted mesh, engineered in an optimal geometric configuration and designed for utmost flexibility while retaining strength characteristics of the fiber material. The embolic protection device is comprised of an ultra-thin polymer mesh protective sleeve, wrapped around the stent. The MGuard(TM) Coronary stent presents a novel combination of a coronary stent merged with an embolic protection device. The GUARDS study will include 60 patients, and its primary endpoints are complete resolution of ST-segment elevation and restoration of flow through the occluded artery. The GUARD Study is a prospective, multi center study, designed to confirm the feasibility, efficacy and safety of MGuard(TM) in acute MI patients. We are confident that the GUARD Study will confirm what we have experienced so far in cath labs around the world that the product performs well and delivers extra safety to acute MI patients." "The successful clinical development of MGuard(TM) will benefit Acute MI patients throughout the world," said Eli Bar, CTO and VP R&D of InspireMD, "MGuard(TM) is designed to provide a comprehensive solution to the embolization threat, during and post procedure. I look forward to confirm these initial findings through this study." From the cases of acute MI where MGuard(TM) was implanted so far in various countries, we learned that the results were very successful and that the product has a strong efficacy and safety profile with an exceptional deliverability. Dariusz Dudek, the GUARD Study Chief Investigator, from Jagiellonian University in Krakow, "The MGuard(TM) represents a very innovative approach to the management of thrombus and embolization, which adversely affect the prognosis and survival rate of a large proportion of Acute MI patients. "It is very exciting for me to see this study take off and confirm our expectations," said Dr. The first 3 patients were implanted successfully with the MGuard(TM) Coronary Stent, in the Jagiellonian University Clinic, in Krakow, Poland. The GUARD Study (MGuard(TM) in Acute Myocardial Infarction Study) is designed to evaluate the safety and efficacy of MGuard(TM) in patients suffering from ST-segment elevation myocardial infarction. announced today the enrollment of the first 3 patients in its study of MGuard(TM) coronary stent in acute myocardial infarction. In the MASTER trial, a total of 433 patients with STEMI presenting within 12 hours of symptom onset undergoing percutaneous coronary intervention were randomized at 50 sites in 9 countries to the MGuard EPS (n = 217) or commercially available bare metal or drug-eluting stents (n = 216).TEL AVIV, Israel, September 23 /PRNewswire/ - InspireMD, Ltd. “The six-month MASTER results highlight the enduring benefits of the MGuard EPS, with a consistent trend in lower mortality.” “The initial MASTER trial results published in the Journal of the American College of Cardiology in October 2012demonstrated the acute benefits of the embolic protection stent, as MGuard EPS outperformed drug-eluting and bare metal stents in complete ST-segment resolution,” said Professor Dr. Sigmund Silber , Director of the Heart Center at the Isar Academic Teaching Site of the University of Munich. Additional 6-month results are available at. These findings corroborate the previously announced 30-day results showing that all-cause mortality with MGuard EPS was lower than bare metal and drug eluting stents used as a control (0% vs. Secondary endpoint clinical outcomes continue to show a lower mortality rate with MGuard EPS compared to control (0.5% vs. The MASTER trial achieved its primary endpoint (p value = 0.008), in complete ST-segment resolution at 60-90 min post-procedure (a strong predictor of mortality). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
